Nucleotides are the basic building blocks of genetics. Thousands of them join together to form DNA (and RNA) molecules. In turn, each nucleotide molecule is itself composed of three subunits: a phosphate group, a five-carbon sugar, and a nitrogen containing base called a nucleobase.
Four nucleobases are found in DNA: adenine (A), cytosine (C), guanine (G), and thymine (T).
As a group, they receive a lot of justifiable attention! But their individuality sometimes gets lost in the larger story of what they accomplish – encoding all the proteins needed for life and enabling DNA to effectively copy and store this information.
Read on to discover a little about what makes each of the ‘fab four of DNA’ unique. Like any good ‘meet the band’article, I promise poster-worthy profile shots and fun trivia. (But no scandals or breakup stories!)
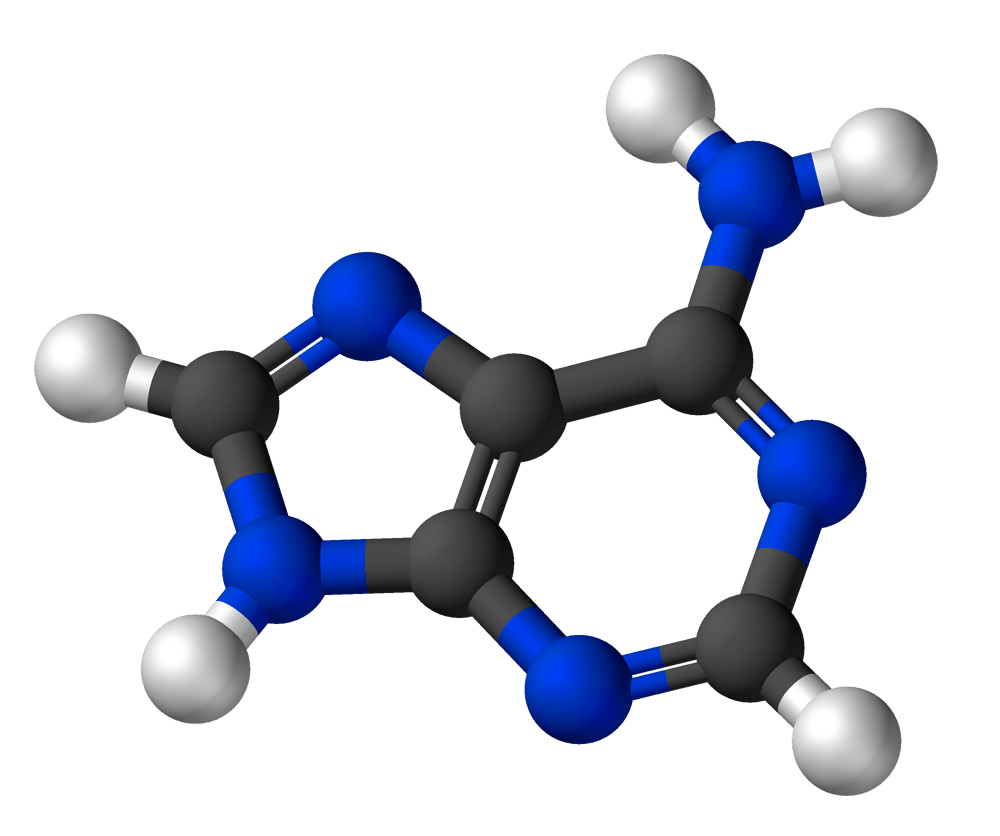
Adenine
Formula: C5H5N5
Molar Mass: 135.13 g/mol
What you may already know: Adenine is one of the two purine bases in the group (the other is guanine). It always pairs with thymine in DNA via two hydrogen bonds. In RNA it binds to another nucleobase – uracil (U).
Multiple Talents: You may not know that adenine also plays a central role in energy storage and cellular respiration. It’s found in several other prominent molecules including adenosine triphosphate (ATP), nicotinamide adenine dinucleotide (NAD), and flavin adenine dinucleotide (FAD).
A Heart Throb: Adenine is also used in medicine. When adenine attaches to a ribose they form adenosine. Adenosine is a prescription drug that can be used to treat paroxysmal supraventricular tachycardia – a heart condition where the patient periodically experiences episodes of rapid heart rate. Adenosine is also part of a cardiac MRI stress test that can help doctors determine if a patient’s heart is receiving enough blood. This diagnostic test is used when the patient is unable to do the more common exercise/treadmill stress test.
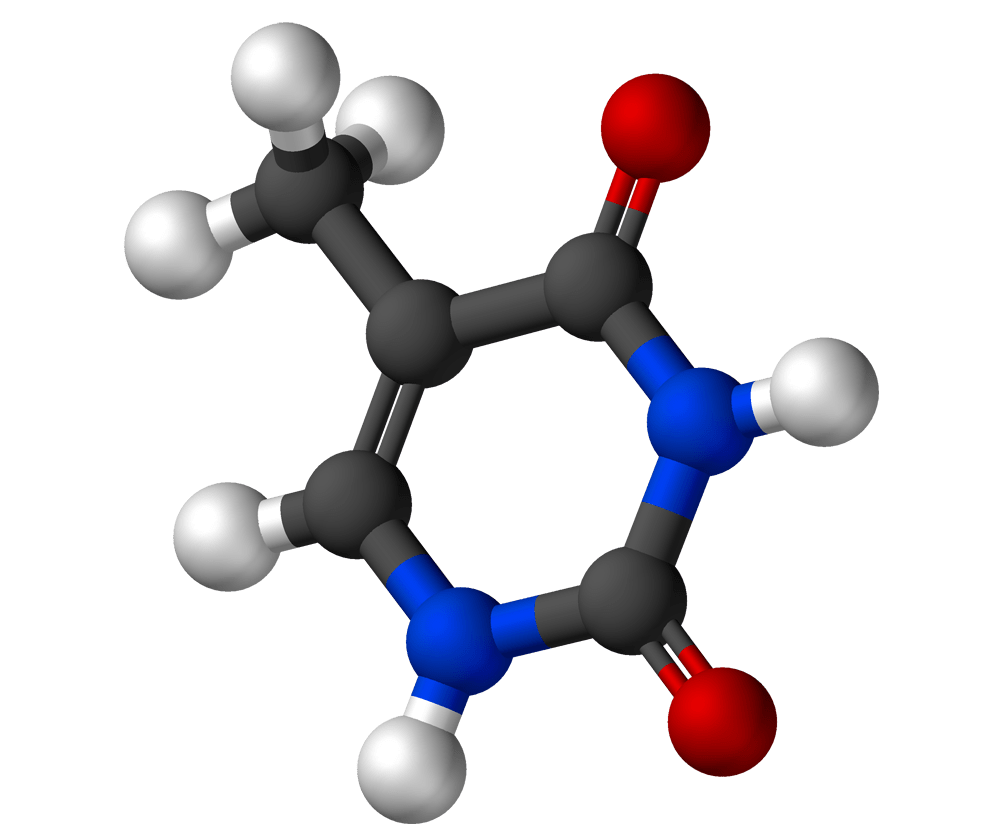
Cytosine
Formula: C4H5N3O
Molar Mass: 110.1 g/mol
What you may already know: Cytosine is one of the two pyrimidine bases in the group (the other is thymine). It always pairs with guanine in DNA via three hydrogen bonds.
Bringing Methyl into the Mix: Methylation occurs when methyl groups (-CH3) attach to DNA and change the activity of that particular stretch of DNA. One of the most common methylation processes is the covalent addition of a methyl group to a cytosine base. This event is so common that the resulting sub-molecule, 5-methylcytosine (5-mC), is sometimes called the “fifth base” of DNA.
A Problem Solver: Quantum computers use ‘qubits’, subatomic particles such as electrons and protons, to solve complex computational problems (instead of the 1 and 0 ‘bits’ used by most common computers). One of the first quantum computers was the Nuclear Magnetic Resonance Quantum Computer (NMRQC). On August 1st, 1998 an Oxford research team used two-qubit NMRQCs based on cytosine molecules to solve David Deutsch’s algorithm. This was the first time ever that quantum mechanical properties were successfully harnessed to process information.
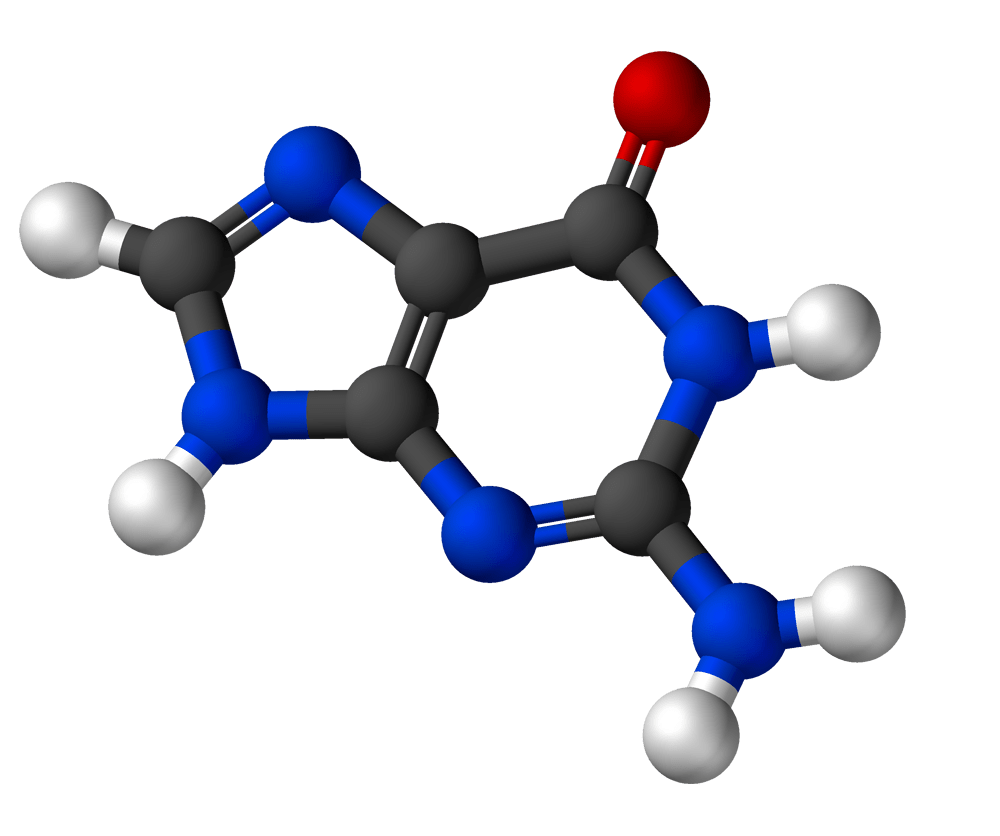
Guanine
Formula: C5H5N5O
Molar Mass: 151.13 g/mol
What you may already know: Guanine is one of the two purine bases in the group. It always pairs with cytosine in DNA via three hydrogen bonds. CG pairs happen to bind more tightly than the AT pairs, so long stretches of CG make stronger helixes than stretches of AT.
What’s in a name?: The discovery of guanine dates back to 1844 when a German scientist named Julius Bodo Unger successfully isolated this molecule in its mineral form from the excreta of sea birds. Two years later it was named guanine from the Spanish word guano which means “bird droppings”.
Favorite look – A pearly luster: Guanine crystalizes into platelets composed of many transparent layers which give the crystals an iridescent appearance. In nature, animals use guanine to both manipulate their appearance (such as chameleons) and to maximize their eyesight (such as crocodiles and deep-sea fish). In industry, guanine is used to create the shimmer often observed in certain paints, eye shadows, nail polishes, and even shampoos.
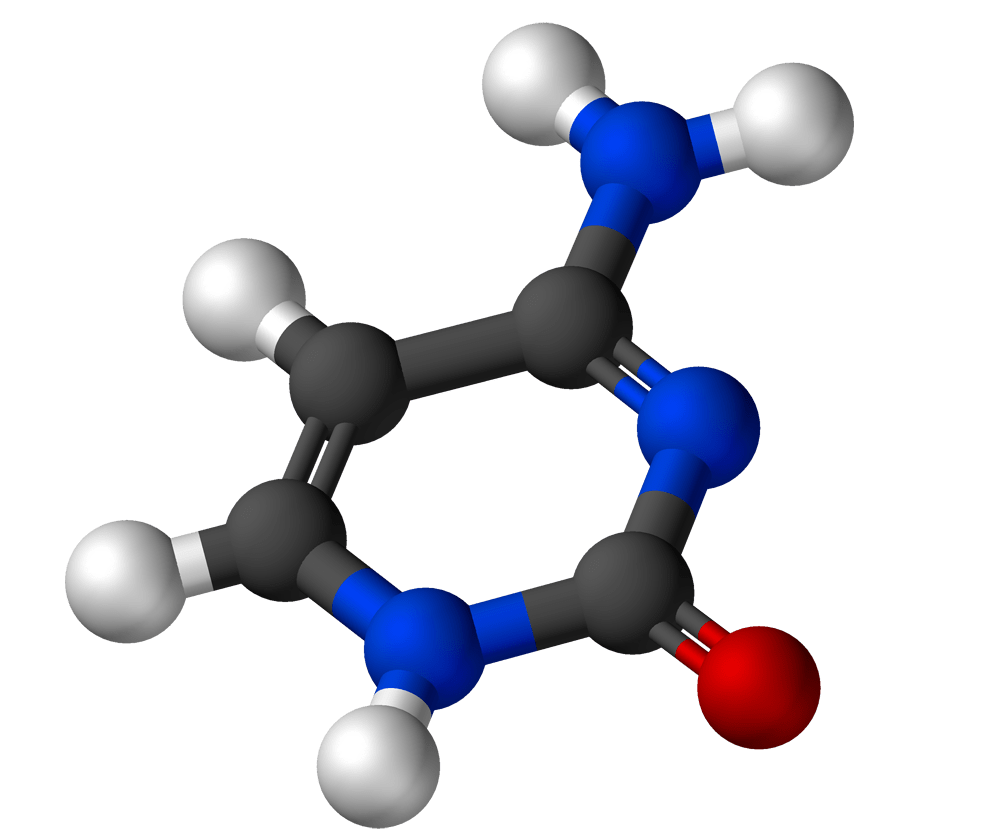
Thymine
Formula: C5H6N2O2
Molar Mass: 126.115 g/mol−
What you may already know: Thymine is one of the two pyrimidine bases in the group. It always pairs with adenine in DNA via two hydrogen bonds.
Prefers cloudy days: One of the most common mutations found in DNA occurs when DNA is exposed to UV radiation and adjected thymines bind to one another forming thymine dimers. These dimers are awkward and change the shape of DNA. These shape changes can cause problems for the enzyme polymerase when the cell needs to replicate its DNA and can sometimes lead to dangerous mutations.
Doubleganger: 5-fluorouracil (5-FU) is a metabolic analog of thymine that can be used in the synthesis of DNA. A commonly used cancer treatment is to introduce 5-FU into actively dividing cancer cells. When these cells incorporate 5-FU into their DNA it can sometimes stop or slow their growth.